- 1Department of Plant Pathology, College of Agriculture, G. B. Pant University of Agriculture and Technology, Pantnagar, India
- 2Academy of Biology and Biotechnology, Southern Federal University, Rostov-on-Don, Russia
- 3ICAR – Central Citrus Research Institute, Nagpur, Nagpur, Maharashtra, India
- 4Division of Bioenergy, Biorefinery and Green Chemistry (BBC-BIC), Department of Energy Technologies and Renewable Resources (TERIN), Italian National Agency for New Technologies, Energy and Sustainable Economic Development (ENEA), Bari, Italy
- 5Faculty of Chemical Sciences, Benemerita, Autonomous University of Puebla, Puebla, Mexico
Complete and balanced nutrition has always been the first line of plant defense due to the direct involvement of mineral elements in plant protection. Mineral elements affect plant health directly by modulating the activity of redox enzymes or improving the plant vigor indirectly by altering root exudates, and changing microflora population dynamics, rhizosphere soil nutrient content, pH fluctuation, lignin deposition, and phytoalexin biosynthesis. Nitrogen (N) is one of the most important macronutrients having a significant impact on the host-pathogen axis. N negatively affects the plant’s physical defense along with the production of antimicrobial compounds, but it significantly alleviates defense-related enzyme levels that can eventually assist in systemic resistance. Potassium (K) is an essential plant nutrient, when it is present in adequate concentration, it can certainly increase the plant’s polyphenolic concentrations, which play a critical role in the defense mechanism. Although no distinguished role of phosphorus (P) is observed in plant disease resistance, a high P content may increase the plant’s susceptibility toward the invader. Manganese (Mn) is one of the most important micronutrients, which have a vital effect on photosynthesis, lignin biosynthesis, and other plant metabolic functions. Zinc (Zn) is a part of enzymes that are involved in auxin synthesis, infectivity, phytotoxin, and mycotoxin production in pathogenic microorganisms. Similarly, many other nutrients also have variable effects on enhancing or decreasing the host susceptibility toward disease onset and progression, thereby making integrative plant nutrition an indispensable component of sustainable agriculture. However, there are still many factors influencing the triple interaction of host-pathogen-mineral elements, which are not yet unraveled. Thereby, the present review has summarized the recent progress regarding the use of macro- and micronutrients in sustainable agriculture and their role in plant disease resistance.
Introduction
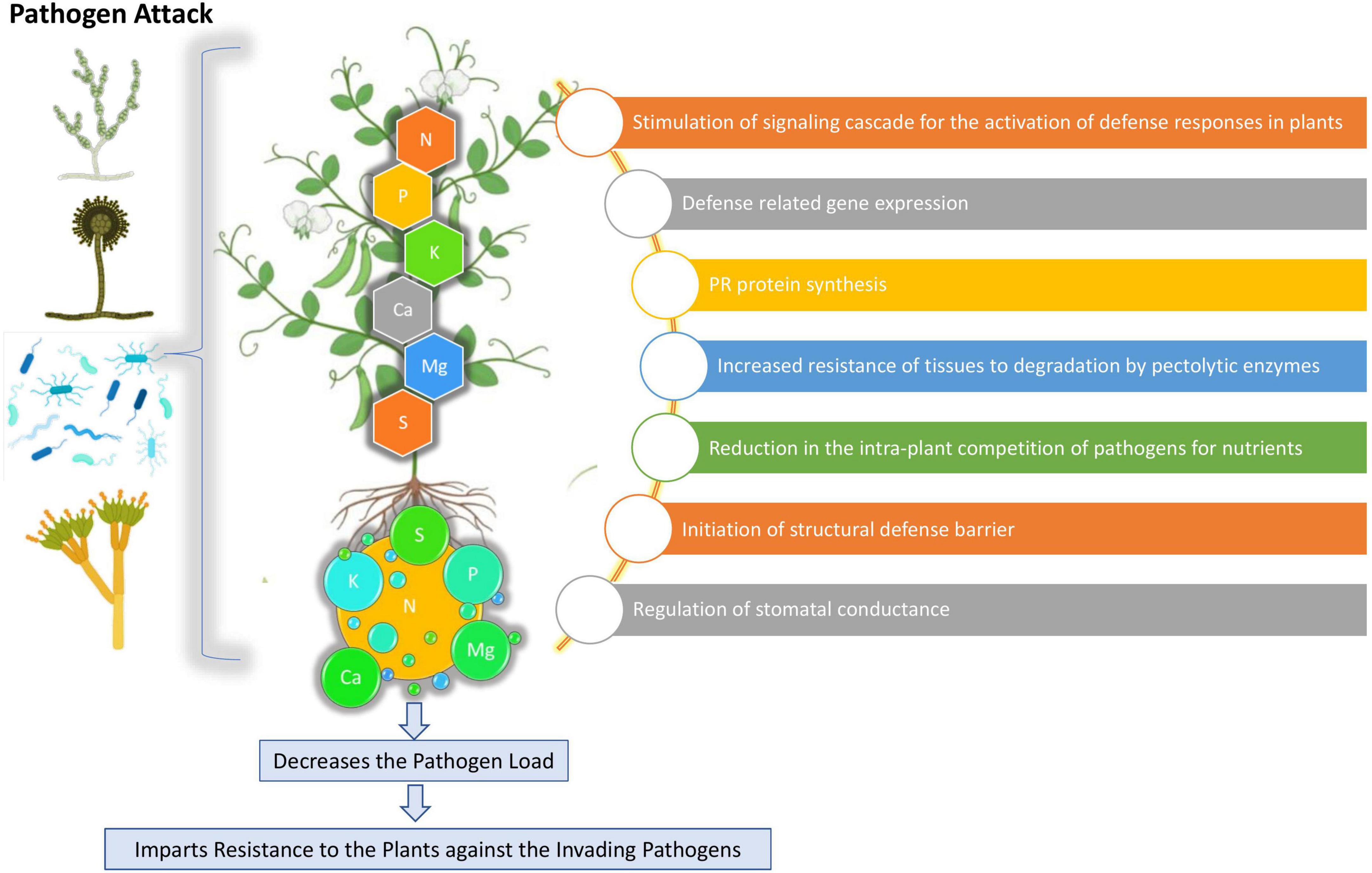
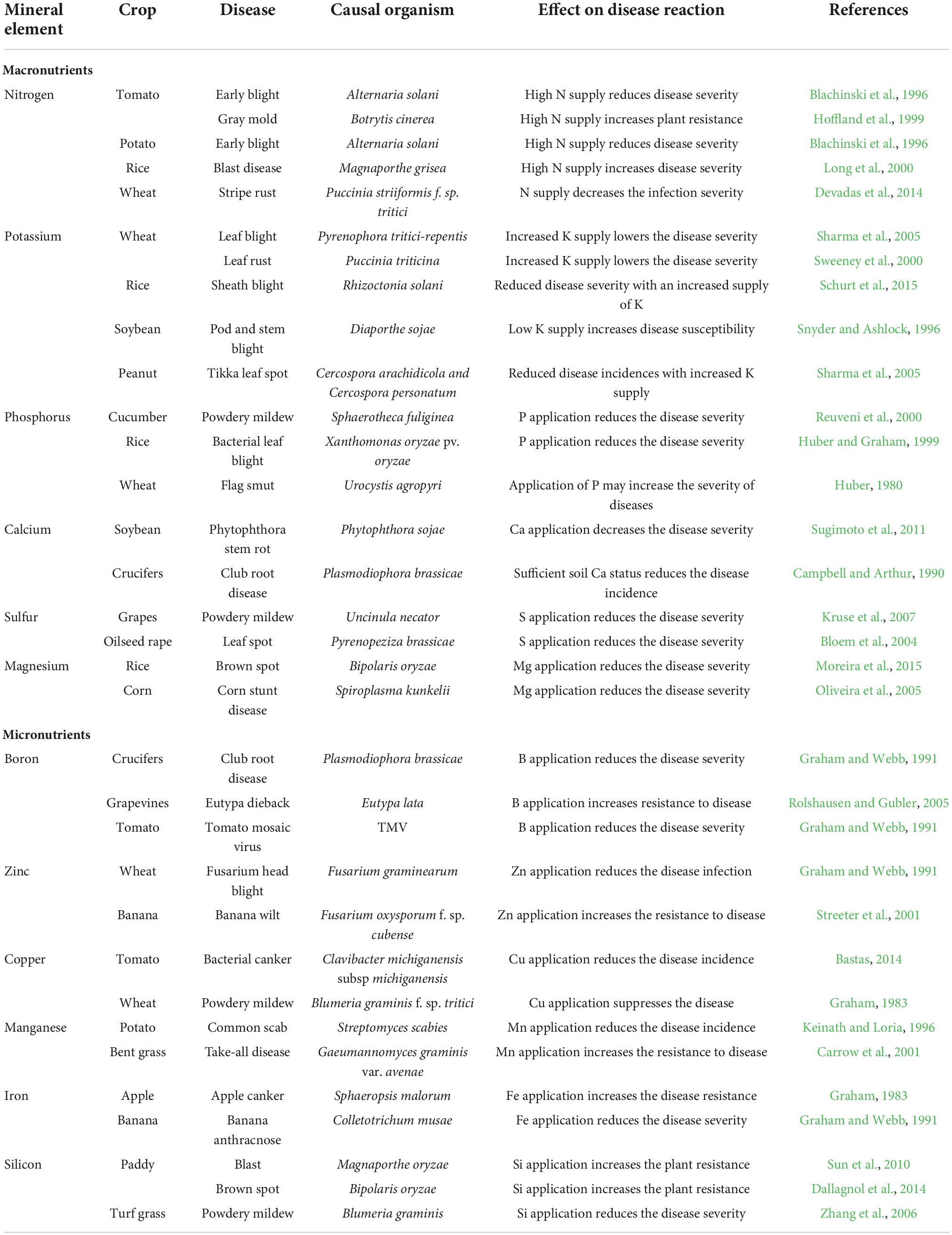
Crop production remains delimited by an array of biotic and abiotic factors that can eventually reduce crop yield, quantity, and quality (Wang M. et al., 2013). Among the biotic factors, phytopathogens such as bacteria, fungi, nematodes, and viruses, have considerable impacts on agricultural productivity and sustainability. Sustainable agriculture can be said as the utilization of the agricultural ecosystem in a way that enables the perfect balance of biological diversity, productivity, and regeneration capacity so that the present and significant future demands can be fulfilled without harming other ecosystems (Lewandowski et al., 1999) and at the same time by managing plant diseases along with an increased yield and improved product quality (Camprubi et al., 2007). Developing along with evolution course, plants have developed multi-layered defense systems enabling them to resist and/or tolerate pathogen invasion and resist infection (Sun et al., 2020). The mineral nutrients play a potential role in supporting plant wellness that is influenced by various abiotic factors, such as light, humidity, temperature, and mineral nutrients (Velasquez et al., 2018) The N status can be affected by high soil temperature as it increases the overall N availability in soils (Lukac et al., 2011) and also increases the plant metabolic rate, thereby positively correlating N uptake with temperature (Dong et al., 2001). The K demands are observed to increase under low moisture conditions, which may sequester a higher reactive oxygen species (ROS) production leading to increased disruption of the plant cell organelles (Wang Y. et al., 2013). The P availability was reported to decrease during high light intensity, which subsequently increased the root length and fine root hair production (Wen et al., 2017). Mineral nutrients are particularly and directly involved in plant protection as structural components and metabolic regulators (Huber, 1980). As imparting the primary defense line, the plant’s nutritional status can play a deciding role in determining the plants’ susceptibility or resistance toward the invading pathogens (Walters and Bingham, 2007; Marschner and Marschner, 2012). The mineral elements can potentially influence plant health either directly by activating the enzymes involved in the synthesis of defense metabolites (callose, glucosinolates, lignin, phenols, and phytoalexins) or indirectly by altering the microbial activity, root exudates composition, and rhizosphere pH modulation (Datnoff et al., 2007). For controlling and managing plant disease, balanced nutrition had always been the primary component, yet its importance remains to be unraveled. The importance of mineral nutrition on plant disease management can be highlighted as (a) fertilization effect on the incidence or severity of a particular pathogen/host pathosystem, (b) mineral nutrition effect in imparting resistance or susceptibility to plant when provided in different concentration, and (c) effect of specific nutrient availability or starvation on disease in consortium with the growth stage of the plant, environmental conditions, and biological activity, which can eventually affect the outcomes (Meena et al., 2015). A healthy plant will certainly have high vigor and improved resistance and hereby mineral nutrients show their capabilities in disease management (Ojha and Jha, 2021). Mineral nutrients, such as the primary macronutrients, nitrogen (N), phosphorus (P), and potassium (K); the three secondary macronutrients, calcium (Ca), sulfur (S), and magnesium (Mg); and the micronutrients, boron (B), manganese (Mn), iron (Fe), zinc (Zn), copper (Cu), and silicon (Si), are significant in imparting disease resistance and healthy growth to the plant (Datnoff et al., 2007; Gupta et al., 2017) (Figures 1, 2). Some key mineral elements have a greater impact on plant disease, for instance, N, which can limit the pathogen growth and may also affect the plant defense elicitation and deployment. Moreover, the availability of different N forms (NH4+ and NO3–) also shows varied effects on plant disease resistance using the assimilatory and metabolic pathways (Bolton and Thomma, 2008; Mur et al., 2017). Similarly, K is particularly a critical element required for plant growth and metabolism and contributes greatly to plants’ survival under various biotic stresses (Pettigrew, 2008) by assisting them in multiple plant defense enzyme functioning, regulating the higher plants’ metabolite patterns, and eventually altering the metabolite concentrations (Mengel, 2001). It can be noted that a particular nutrient may have opposite impacts on different diseases and in different environments, i.e., the same nutrient may increase the incidence of one disease but at the same time may decrease the incidence of others (Agrios, 2005) (Table 1).
This review presents the recent advances bridging the implications of mineral nutrients in sustaining plant health, with a focus on nutrient signaling and disease resistance. In addition, an attempt has been made to unravel the linkage between plant macro/micronutrients involved in the disease onset and progression, thereby ensuring sustainable crop production.
Macronutrients mediated plant disease management
Nitrogen
In the context of plant disease management, nitrogen (N) is an essentially important macronutrient required for the normal growth and development of the plant (Scheible et al., 2004). N plays a prominent role in varying metabolic and physiological processes, such as photosynthesis, amino acid synthesis, respiration, and tricarboxylic acid (TCA) cycle (Foyer et al., 2011). The N availability can restrict pathogen growth by alleviation and deployment of different plant defense mechanisms, and the different forms of N (NH4+ and NO3 form) are reported to have diverse effects on plant disease resistance (Bolton and Thomma, 2008; Mur et al., 2017). Several instances have been reported wherein N fertilization increased the plant disease incidence, for example, downy mildew, powdery mildew, leaf rust, stem rot, and rice blast diseases (Ballini et al., 2013; Devadas et al., 2014; Huang et al., 2017) while contrary results have been reported for diseases, such as take-all, gray mold, and leaf spot (Krupinsky et al., 2007; Lecompte et al., 2010). The excessive use of N fertilization in plants promotes succulent tissue growth and alleviates apoplastic amino acid concentration along with improving the plant canopy, which ultimately favors the growth of pathogenic spores (Neumann et al., 2004; Dordas, 2008).
The impact of N limitation on Pseudomonas syringae pv. syringae B728a when studied through an extensive transcriptomic assessment revealed the prominence of virulence-associated features, such as swarming motility, type three secretion system (T3SS), and metabolic pathways involved in gamma-aminobutyric acid (GABA) and polyketide metabolism (Bolton and Thomma, 2008). N starvation studies confirm its importance in initiating pathogenesis by stimulating the pathogen effector genes, such as the hypersensitive response and pathogenicity (hrp), avirulence (avr), and hydrophobin MPG1 genes in Magnaporthe oryzae (Pérez-García et al., 2001) while opposite results were documented for effectors from Magnaporthe oryzae (Huang et al., 2017) and Passalora fulva (ex Cladosporium fulvum) (Thomma et al., 2005). Defense enzymes are also an important arsenal possessed by plants in fighting the invading pathogen and N is observed to be involved in the stimulation of these enzymes during the host-pathogen interaction (Ngadze et al., 2012). The genes encoding the key regulatory enzymes of the defense pathway, such as phenyl ammonia lyase (PAL), cinnamate-4-hydroxylase (C4H), and 4-coumarate: CoA ligase (4CL), are all upregulated by N deficiency (Camargo et al., 2014) while a reduction in PAL activity has been observed with N fertilization (Sun et al., 2018). However, the relationship between N fertilization and plant disease is still unclear, but the understanding of the fundamental mechanism is noteworthy in crop production.
Phosphorus
Phosphorus (P) is thought to be the second most commonly applied nutrient after nitrogen in crops but its role in resistance is seemingly inconsistent and variable. P is a part of many cell organic + molecules, such as deoxyribonucleic acid (DNA), ribonucleic acid (RNA), adenosine triphosphate (ATP), and is also involved in many metabolic processes taking place both in the plant and in the pathogen. During pathogen infection, the extracellular ATP is also received as a damage-associated molecular pattern (DAMP) since it is sensed by the plant when cellular damage is caused during pathogen colonization (Tanaka et al., 2014), considering it as a signaling molecule for the defense response activation in the plant (Cao et al., 2014). In recent reports, the role of extracellular ATP has been also proposed in jasmonic acid (JA)-induced defense response through direct activation of JA signaling in the Arabidopsis plant (Tripathi et al., 2018; Jewell et al., 2019). The beneficial effects of P application are also observed in controlling seedling and fungal diseases wherein the prolific root growth enables the plant to escape the disease (Huber and Graham, 1999). Various researchers have shown the significant effect of P fertilization in managing Pythium root rot in wheat (Huber, 1980) and reducing bacterial leaf blight in rice, downy mildew, blue mold, leaf curl virus disease in tobacco, pod and stem blight in soybean, yellow dwarf virus disease in barley, brown stripe disease in sugarcane, and blast disease in rice (Potash and Phosphate Institute [PPI], 1988; Reuveni et al., 1998; Huber and Graham, 1999; Kirkegaard et al., 1999; Reuveni et al., 2000). Campos-Soriano et al. (2020) reported overexpression of miR399 resulting in high Pi content and enhanced susceptibility to infection by the rice blast fungus Magnaporthe oryzae due to high phosphate fertilization.
Potassium
Potassium (K) is an essential nutrient and the most plentiful inorganic cation found in plants (Shabala and Pottosin, 2010). K plays essential roles in enzyme activation, protein synthesis, photosynthesis, osmoregulation, stomatal movement, energy transfer, phloem transport, cation-anion balance, stress resistance (Marschner, 2012) crop yield, and quality improvement (Marschner, 2012; Oosterhuis et al., 2014). The plants with K starvation symptoms are observed to be more susceptible to disease in comparison to those having adequate K supply. A reduction in the incidence of fungal diseases (70%), bacterial diseases (69%), viral diseases (41%), and nematodes (33%) due to the profound K use was reported by Perrenoud (1990). Though K fertilization decreased the disease incidence in most of the cases, contrary results were also reported in some instances thereby categorizing the K impact on plant disease as “increased,” “decreased,” and having “no effect” or “variable effect” (Prabhu et al., 2007). The increased susceptibility in strawberries grown under K concentration excess toward Colletotrichum gloeosporioides and resistance alleviation in K fertilization absence due to starvation-induced synthesis of ROS and phytohormones were reported by Nam et al. (2006) which lead to enhanced plant stress tolerance (Amtmann et al., 2008). The increased K+ concentrations also decrease the prevailing intra-plant pathogen competition for nutrients (Holzmueller et al., 2007) and thereby enabling the plant to divert more resources to build the physical defense barrier and damage repair (Mengel, 2001). K is also an important facet in regulating the plant enzyme function by regulating the plants’ metabolite pattern and eventually varying its metabolite concentrations (Marschner, 2012). The synthesis of high-molecular-weight compounds (such as proteins, starches, and cellulose) and phenol concentration was significantly increased in plants with an adequate supply of K, which depressed the concentrations of low-molecular-weight compounds (soluble sugars, organic acids, amino acids, and amides) essential for diseases development in plant tissues, thereby making the plant less prone toward disease incidence (Prasad et al., 2010).
Calcium
Calcium is an essential element, serving as one of the cell wall and membrane constituents and thereby contributing to the cell structure along with upholding the physical barriers against invading pathogens (White and Broadley, 2003). Owing to its significance in the structural role, the plants showing Ca deficiency are observed to be more prone to disease infection, and element exogenous supply has been shown to alleviate the plant’s resistance response toward the pathogen. A reduction in the Ca concentration within the plant increases susceptibility toward the fungi preferentially invading the xylem tissue and dissolving the cell wall of the conducting vessels increases, leading to wilting of the plant (Hirschi, 2004). In addition, Ca also plays an important role in serving as a secondary messenger for a variety of metabolic processes carried out within the plant during biotic stresses (Lecourieux et al., 2002). The Ca2+ signal is observed to be one of the earliest responses in the basal defense response triggering the signaling cascade required for the pathogen-associated molecular patterns (PAMPs) or host-derived damage-associated molecular patterns (DAMPs) that are recognized by surface-localized pattern-recognition receptors (PRRs) eventually leading to PAMP-triggered immunity (PTI) (Dodds and Rathjen, 2010).
Sulfur
Sulfur (S) is an essential plant macronutrient having a pivotal role in plant disease resistance. The sulfur-containing defense compounds (SDCs) play versatile roles both in pathogen perception and initiating signal transduction pathways that are interconnected with various defense processes regulated by plant hormones (salicylic acid, JA, and ethylene) and ROS (Kunstler et al., 2020). The sulfur-containing amino acid (SAA) cysteine acts as a precursor of a large number of biomolecules, having major roles in plant disease resistance. Cysteine mediates spore germination and mycelial growth inhibition in a concentration-dependent manner in Phaeomoniella chlamydospora and Phaeoacremonium minimum, the two main causal agents of grapevine trunk disease (Roblin et al., 2018). The other important SAA in plants playing a central role in different defense reactions to biotic stresses is methionine (Met). A drastic reduction in the disease severity of Met-treated susceptible pearl millet cultivar (Pennisetum glaucum) infected by Sclerospora graminicola was reported by Sarosh et al. (2005). The Met treatment induces the generation of hydrogen peroxide (H2O2), a key element in plant defense signaling, leading to an upregulation in different defense-related gene expressions in grapevine (Vitis vinifera) (Boubakri et al., 2013). Sulfur-containing secondary metabolites play an important role in plant disease resistance and based on their mode of action can be classified into phytoalexins and phytoanticipins (Nwachukwu et al., 2012). In sulfur-deficient plants, there is a general gene downregulation responsible for sulfur-containing secondary metabolites synthesis and therefore the biosynthesis of S-containing phytoalexin (Camalexin) is also inhibited. Elemental sulfur (S0) can also be regarded as the only inorganic phytoalexin in plants that is accumulated during the xylem-invading fungal infection and bacterial pathogens infection, and its accumulation is faster and greater in disease-resistant genotypes than in susceptible lines (Cooper and Williams, 2004). The reactive sulfur species (RSSs) also play an important role in defense metabolism due to their participation in cellular signaling and regulatory processes. Two RSSs, hydrogen sulfide and sodium sulfite, have been shown to play important roles in plant disease resistance (Gao et al., 2012; Chen et al., 2014).
Magnesium
Magnesium (Mg) is a vital cation, which influences an array of in planta physiological functions when the plant presents deficient or excess concentrations (Wang et al., 2020). It can also affect the pathogen invasion way into a plant by colonizing the plant phloem tissues, as it is present within the young phloem tissues under high Mg concentration and outside the cells under Mg deficit conditions. A low Mg concentration was detected in maize plants infected with corn stunt spiroplasma, which occurs due to the competition for Mg between the plant and the pathogen, thereby causing pronounced symptoms in the plant deficient in Mg (Nome et al., 2009). Mg deficiency during plant growth can also reduce the structural integration within the middle lamella and may also lower the energy production necessary for defense functions eventually leading to pathogen metabolites inactivation. A nutrient-rich environment favoring several phytopathogens occurs in the leaf tissue under the Mg deficiency condition due to sucrose and starch deposition in the leaf tissue (Huber and Jones, 2013). A higher clubroot disease incidence was also reported in soils showing lower Mg concentrations (Young et al., 1991). A drastic increase in the rate of disease infection and severity of peanut leaf spots caused by Mycosphaerella arachidicola was observed during the Mg deficient conditions (Bledsoe et al., 1945). An increase in pepper and tomato bacterial spot disease incidence caused by Xanthomonas campestris pv. vesicatoria was observed due to alleviated Mg levels (Woltz and Jones, 1979).
Micronutrients mediated plant disease management
Boron
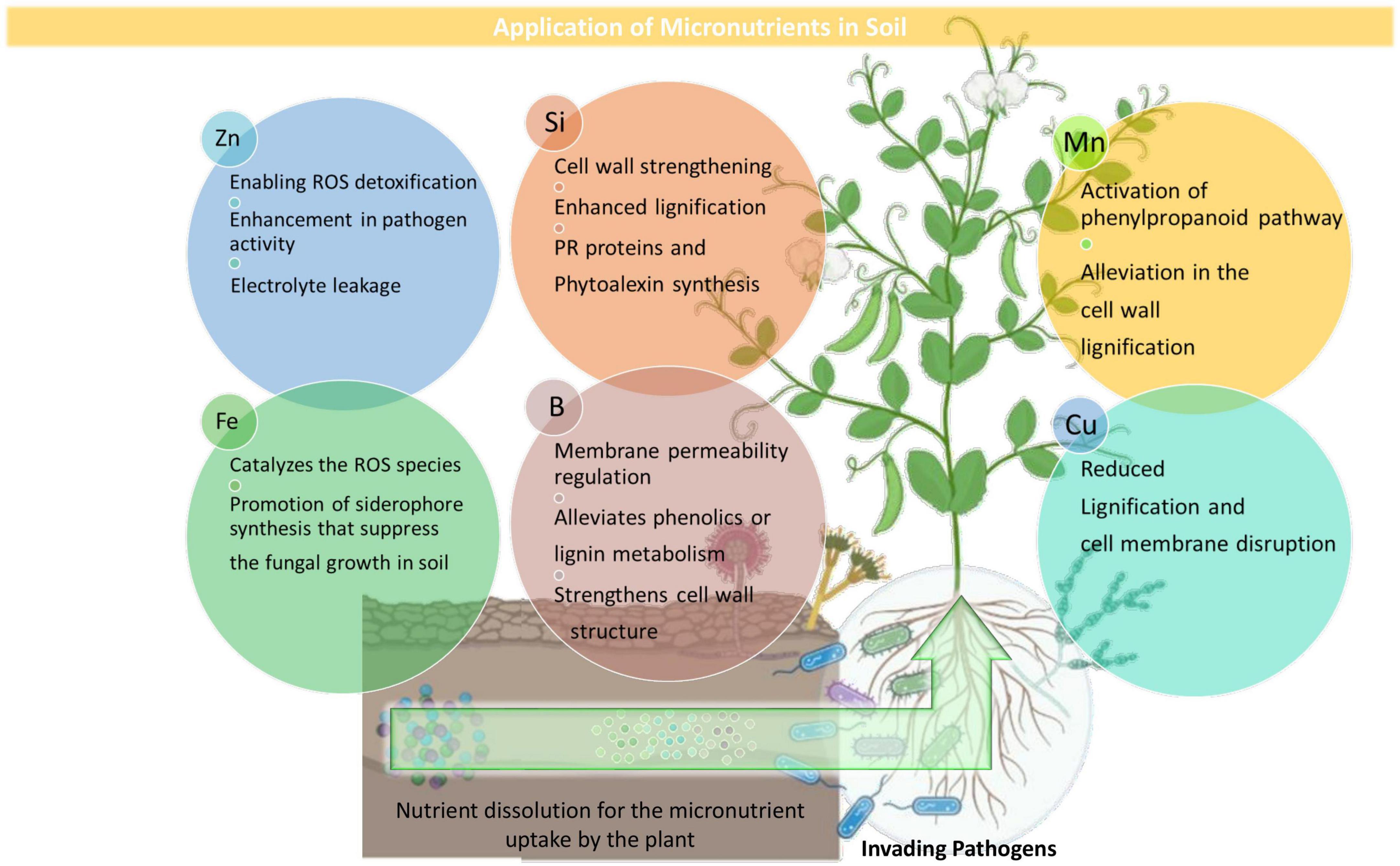
Boron (B) is one of the least understood micronutrients showing widespread deficiency in plants around the globe (Brown et al., 2002). B nutrition-mediated physiological and metabolic activities that reduce disease susceptibility in the plant system are attributed to (1) strengthening cell wall structure through the formation of carbohydrate-borate complexes, which control carbohydrate transport and cell wall protein metabolism, (2) controlling cell membrane permeability and stability function, and (3) phenolics or lignin metabolism (Brown et al., 2002). In B deficient conditions, plant cell walls tend to swell and split, resulting in weakened intercellular space, which eventually weakens the physical barrier to the initial infection (Blevins and Lukaszewski, 1998). Sanjeev and Eswaran (2008) observed that B nutrition contributed to the maximum fungal mycelial growth inhibition and it can be used as a prokaryotic inhibitor at a certain concentration. The response of soil-borne phytopathogenic prokaryotes, such as Ralstonia, Pectobacterium, and Pantoea, to B can be assessed, and if boron concentration is not toxic to other beneficial plant-associated microorganisms, then altered B nutrition can be used as disease management effective means against the soil-borne plant pathogens.
Zinc
Zinc (Zn), one of the crucial micronutrients, plays its role in plant response toward phytopathogens primarily activating or stabilizing metalloenzymes (Fones and Preston, 2012). Generally, the Zn deficient plants are more prone to pathogen attack (Streeter et al., 2001), thereby providing Zn the status of a significant element deciding the outcome of the plant-pathogen interaction. This results in limiting the invader’s entry or evading plant defense responses. Several studies suggest Zn fertilization role in reducing plant symptoms (Li et al., 2016; Machado et al., 2018); however, an increased susceptibility toward other pathogens was also reported due to protective Zn concentrations used against certain pathogens of the same host (Helfenstein et al., 2015). The studies conducted on the potential relationship between Zn availability status and fungal disease severity have reported an alleviated disease response in plants supplemented with Zn (Huber and Haneklaus, 2007; Khoshgoftarmanesh et al., 2010) while the contrary results were observed in soybean plants with varied Zn treatment, wherein either normal or high Zn fertilization had fewer positive counts for bacterial pustules caused by Xanthomonas axonopodis pv. glycines (Helfenstein et al., 2015). An evolutionary-conserved Zn-sensing phenomenon connecting root growth to pathogen response mechanism was stated by Bouain et al. (2018). In this study, the authors found that azelaic acid triggered by Azelaic Acid Induced1 (AZI1), belonging to the lipid transfer protein family (LTP) of the pathogenesis-related (PR) protein during systemic acquired resistance (SAR), regulated the plant growth and immunity responses on the basis of Zn availability status in plants.
Copper
Copper (Cu) is one of the significant micronutrients required by plants that acts as a cofactor for several enzymes involved in respiration and electron transport proteins (Sommer, 1931). Cu is a plant protection essential part of controlling oomycetes, fungi, and bacteria for over a century. Although diseases can be managed by Cu applications, the lack of curative or systemic activity leads to Cu spray applications year after year (Graham et al., 2011). Plants with low Cu content show an increased disease incidence as a result of reduced lignification (Marschner, 1995). Cu fertilization in plants reduces the severity of fungal and bacterial diseases associated with cell wall stability and lignification (Broadley et al., 2012). The best evidence of a Cu effect on host plant resistance to disease can be observed in cases where Cu is applied in soil, and it lowers the leaf infection as evident in powdery mildew in wheat and ergot (Claviceps sp.) (Evans et al., 2007). The Cu synergistic effects can also be stated when it was used with other fungicides, such as Mancozeb, which leads to a reduction in canker and fruit spotting symptoms (Shoemaker, 1992). Cu compounds and their different combinations, in different studies, are reported to reduce sheath blight severity (Rhizoctonia solani) in rice (Khaing et al., 2014) and bacterial canker (Clavibacter michiganensis subsp. michiganensis) in tomatoes (Bastas, 2014).
Manganese
Manganese (Mn) is an important micronutrient known for its efficacy on pathogen and resistance development in plants (Huber and Graham, 1999) owing to its ability for phenolic and lignin compound synthesis (Broadley et al., 2012). Cacique et al. (2012) reported that high Mn concentration on leaf tissues was found to decrease blast symptoms by Pyricularia oryzae in rice. Heine et al. (2011) observed that Mn can also contribute to black leaf mold disease control (Pseudocercospora fuligena) in tomatoes. Plants with inadequate Mn nutrition are observed to be unable in restricting the fungal hyphae penetration into the root tissues (Graham and Webb, 1991) while plants with adequate Mn nutrition show an alleviation in lignification and a reduction in aminopeptidase and pectin methyl esterase synthesis that is required essentially for fungal growth and for host cell wall breakdown, respectively (Carrow et al., 2001).
Iron
Iron (Fe) is an essential micronutrient required by plants and pathogens having both positive and negative effects on the host and host disease resistance (Kieu et al., 2012; Aznar et al., 2015). Fe catalyzes ROS production that is used by the plant for alleviating the local oxidative stress as a defense response against the pathogens, thereby making iron play an intricate role in plant-pathogen interaction (Aznar et al., 2015). Fe fertilization is evident to be effective in antimicrobial compound synthesis leading to an indirect effect on the plant’s metabolic activity (Aznar et al., 2015). A reduction in symptom severity and pectate lyase encoding gene expression of the two soft rot-causing pathogens, such as Dickeya dadantii and Botrytis cinerea, was observed in plants showing Fe starvation (Kieu et al., 2012). Fe is also reported to enhance the fungal growth in certain plant-fungus interactions, as it was observed in Phytophthora parasitica var. nicotianae, wherein the fungal growth was observed to enhance in Fe3+ supplemented synthetic glucose asparagine medium (Hendrix et al., 1969). Fe also plays a potent role in Pseudomonads that are adapted to produce iron-chelating agents called siderophores in Fe-deficient soils, which, in turn, suppress certain fungal pathogens by starving them of iron (Calvent et al., 2001). Siderophores are also involved in some volatile antibiotic compounds’ synthesis (Thomashow, 1996). Depending on the host, the defense activation mechanism involves either their Fe scavenging property or receptor-mediated recognition as in the case of pattern-triggered immunity (Aznar and Dellagi, 2015). The reduced iron availability for fusaria-related wilts induced by fluorescent pseudomonads producing siderophores is reported as the main mechanism to reduce disease incidence in fusarium wilt of tomatoes (De Weger et al., 1986; Alabouvette, 1999; Hussain et al., 2016). In fact, soil suppressiveness to fusarium wilt of tomatoes has been mainly ascribed to Fe competition between the pathogenic Fusarium oxysporum isolates from the rhizosphere with the wild populations of fluorescent pseudomonads (Haas and Défago, 2005; Lemanceau and Alabouvette, 2008).
Silicon
Silicon (Si) is not essentially a micronutrient but stands out eminently in its potential for decreasing several pathogens’ severity in varied crops belonging to the families Poaceae, Equisetaceae, and Cyperaceae (Huber et al., 2012; Pozza et al., 2015). The increased Si supply strongly reduces the number of lesions on young leaves, indicating an increase in disease resistance (Osuna-Canizalez et al., 1991). The silicates are known for inducing defense responses in plants by involving cell wall strengthening through alleviated phytoalexin production, increased lignification, PR protein synthesis, and phenolics production (Fawe et al., 2001; Oliveira et al., 2012).
Silicon is accumulated mainly in epidermal cells and exclusively on endodermal cells in roots and creates a physical barrier for fungal hyphae penetration into plant roots (Najihah et al., 2015). An increase in the activity of antioxidative enzymes (peroxidase, polyphenol oxidase, phenylalanine ammonia lyase, and lipoxygenase) was also observed after Si application (Shetty et al., 2011), which are considered the second line of defense for the pathogen entry into the host (Pozza et al., 2015). A significant reduction in lesion length of bacterial leaf blight (Xanthomonas oryzae pv. oryzae) among four rice cultivars was reported by Chang et al. (2002) following Si application, which was correlated with the soluble sugar content reduction in plant leaves amended with Si. Reduced severity in rice sheath blight disease was attributed to the increased lignin content and enhanced activities of antioxidative enzymes in rice leaves with Si addition. Therefore, knowing its effects on disease reduction, it can be included as an important component of crop protection.
Conclusion
Since sustainable agriculture that uses increasing amounts of bio-fertilizers and organic amendments from a wide range of organic wastes represents a very important plant mineral nutrient source, it is fundamental to know the mechanisms of action by which such minerals can be involved in plant defense in several pathosystems. It is a general assumption that balanced nutrition leads to a healthy plant, which reduces disease susceptibility and infection. Thus, it is important to provide balanced nutrition at the due time when the nutrient can be most effectively used for disease control. Nutrient manipulation achieved by either modifying the nutrient availability or modifying the nutrient uptake for disease management or suppression has been reported in several studies. Fertilizers’ application affects plant disease development under field conditions either directly through the plant’s nutritional status or indirectly by affecting the conditions, which can influence the disease development, such as dense stands, changes in light interception, and humidity within the crop stand. It is a general assumption that balanced nutrition leads to a healthy plant, which reduces disease susceptibility and infection. Thus, it is important to provide balanced nutrition at the due time when the nutrient can be most effectively used for disease control.
Author contributions
KPS provided the concept for the manuscript. RTr, RTe, and CK drafted the original manuscript. UDC, ES, TM, and AKS edited and supervised the final manuscript. All authors contributed to the article and approved the submitted version.
Acknowledgments
RrT, RTe, and KPS are jointly grateful to G. B. Pant University of Agriculture and Technology for meeting the necessary fund requirement.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Alabouvette, C. (1999). Fusarium wilt suppressive soils: An example of disease-suppressive soils. Australas. Plant Pathol. 28, 57–64. doi: 10.1071/AP99008
Amtmann, A., Troufflard, S., and Armengaud, P. (2008). The effect of potassium nutrition on pest and disease resistance in plants. Physiol. Plantarum. 133, 682–691. doi: 10.1111/j.1399-3054.2008.01075.x
Aznar, A., Chen, N. W. G., Thomine, S., and Dellagi, A. (2015). Immunity to plant pathogens and iron homeostasis. Plant Sci. 240, 90–97. doi: 10.1016/j.plantsci.2015.08.022
Aznar, A., and Dellagi, A. (2015). New insights into the role of siderophores as triggers of plant immunity: what can we learn from animals? J. Exp. Bot. 66, 3001–3010. doi: 10.1093/jxb/erv155
Ballini, E., Nguyen, T. T., and Morel, J. B. (2013). Diversity and genetics of nitrogen-induced susceptibility to the blast fungus in rice and wheat. Rice 6:32. doi: 10.1186/1939-8433-6-32
Bastas, K. K. (2014). Effects on tomato bacterial canker of resistance inducers and copper compound in greenhouse. J. Agri. Food Sci. 28, 1–10.
Blachinski, D., Shtienberg, D., Dinoor, A., Kafkafi, U., Sujkowski, L. S., Zitter, T. A., et al. (1996). Influence of foliar application of nitrogen and potassium on Alternaria diseases in potato, tomato and cotton. Phytoparasitica 24, 281–292. doi: 10.1007/BF02981411
Bledsoe, R. W., Harris, H. C., and Tisdale, W. B. (1945). Leafspot of peanut associated with magnesium deficiency. Plant Physiol. 21, 237–240. doi: 10.1104/pp.21.2.237
Blevins, D. G., and Lukaszewski, K. M. (1998). Boron in plant structure and function. Ann. Rev. Plant Physiol. 49, 481–500. doi: 10.1146/annurev.arplant.49.1.481
Bloem, E., Riemenschneider, A., Volker, J., Papenbrock, J., Schmidt, A., Salac, I., et al. (2004). Sulphur supply and infection with Pyrenopeziza brassicae influence L-cysteine desulphydrase activity in Brassica napus L. J. Exp. Bot. 55, 2305–2312. doi: 10.1093/jxb/erh236
Bolton, M. D., and Thomma, B. P. H. J. (2008). The complexity of nitrogen metabolism and nitrogen-regulated gene expression in plant pathogenic fungi. Physiol. Mol. Plant Pathol. 72, 104–110. doi: 10.1016/j.pmpp.2008.07.001
Bouain, N., Satbhai, S. B., Korte, A., Saenchai, C., Desbrosses, G., Berthomieu, P., et al. (2018). Natural allelic variation of the AZI1gene controls root growth under zinc-limiting condition. PLoS Gen. 14:e1007304. doi: 10.1371/journal.pgen.1007304
Boubakri, H., Wahab, M. A., Chong, J., Gertz, C., Gandoura, S., Mliki, A., et al. (2013). Methionine elicits H2O2 generation and defense gene expression in grapevine and reduces Plasmopara viticola infection. J. Plant Physiol. 170, 1561–1568. doi: 10.1016/j.jplph.2013.06.008
Broadley, M., Brown, P., Cakmak, I., Rengel, Z., and Zhao, F. (2012). “Function of nutrient: micronutrients,” in Marschner’s Mineral Nutrition of Higher Plants, ed. P. Marschner (Sydney: Academic Press), 191–248. doi: 10.1016/B978-0-12-384905-2.00007-8
Brown, P. H., Bellaloui, N., Wimmer, M. A., Bassil, E. S., Ruiz, J., Hu, H., et al. (2002). Boron in plant biology. Plant Biol. 4, 205–223. doi: 10.1055/s-2002-25740
Cacique, I. S., Domiciano, G. P., Rodrigues, F. A., and do Vale, F. X. R. (2012). Silicon and manganese on rice resistance to blast. Bragantia 71, 239–244. doi: 10.1590/S0006-87052012000200013
Calvent, V., de Orellano, M. E., Sansone, G., Benuzzi, D., and de Tosetti, M. I. S. (2001). Effect of nitrogen source and pH on siderophore production by Rhodotorula strains and their application to biocontrol of phytopathogenicmoulds. J. Ind. Microbiol. Biotech. 26, 226–229. doi: 10.1038/sj.jim.7000117
Camargo, E. L. O., Nascimento, L. C., Soler, M., Salazar, M. M., Lepikson-Neto, J., Marques, W. L., et al. (2014). Contrasting nitrogen fertilization treatments impact xylem gene expression and secondary cell wall lignification in Eucalyptus. BMC Plant Biol. 14:256. doi: 10.1186/s12870-014-0256-9
Campbell, N., and Arthur, S. G. (1990). “Control of clubroot of crucifers by liming,” in Management of Diseases With Macro- and Microelements, ed. A. W. Engelhard (Paul, MN: APS Press), 90–101.
Campos-Soriano, L., Bundó, M., Bach-Pages, M., Chiang, S. F., Chiou, T. J., and San Segundo, B. (2020). Phosphate excess increases susceptibility to pathogen infection in rice. Mol. Plant Pathol. 21, 555–570. doi: 10.1111/mpp.12916
Camprubi, A., Estaun, V., El Bakali, M. A., Garcia-Figueres, F., and Calvet, C. (2007). Alternative strawberry production using solarization, metham sodium and beneficial soil microbes as plant protection methods. Agron. Sustain. Dev. 27, 179–184. doi: 10.1051/agro:2007007
Cao, Y., Tanaka, K., Nguyen, C. T., and Stacey, G. (2014). Extracellular ATP is a central signaling molecule in plant stress responses. Curr. Opin. Plant Biol. 20, 82–87. doi: 10.1016/j.pbi.2014.04.009
Carrow, R. N., Waddington, D. V., and Rieke, P. E. (2001). Turfgrass Soil Fertility and Chemical Problems: Assessment and Management. New York, NY: Ann Arbor Press.
Chang, S. J., Tzeng, D. D. S., and Li, C. C. (2002). “Effect of silicon nutrient on bacterial blight resistance of rice (Oryza sativa L.),” in 2nd Silicon in Agriculture Conference, ed. T. Matoh (Kyoto: Press-Net), 31–33.
Chen, J., Liu, T. W., Hu, W. J., Simon, M., Wang, W. H., Chen, J., et al. (2014). Comparative proteomic analysis of differentially expressed proteins induced by hydrogen sulfide in Spinacia oleracea leaves. PLoS One 9:e105400. doi: 10.1371/journal.pone.0105400
Cooper, R. M., and Williams, J. S. (2004). Elemental sulphur as an induced antifungal substance in plant defense. J. Exp. Bot. 55, 1947–1953. doi: 10.1093/jxb/erh179
Dallagnol, L. J., Rodrigues, F. A., Mielli, M. V. B., and Ma, J. F. (2014). Rice grain resistance to brown spot and yield are increased by silicon. Tropic Plant Pathol. 39, 56–63. doi: 10.1094/PHYTO-99-1-0116
Datnoff, L. E., Elmer, W. H., and Huber, D. M. (2007). Mineral Nutrition and Plant Disease. St. Paul Davis: APS Press.
De Weger, L., Van Boxtel, R., Van der Burg, B., Gruters, R., Geels, F., Schippers, B., et al. (1986). Siderophores and outer membrane proteins of antagonistic, plant-growth-stimulating, root-colonizing Pseudomonas sp. J. Bacteriol. 165, 585–594. doi: 10.1128/jb.165.2.585-594.1986
Devadas, R., Simpfendorfer, S., Backhouse, D., and Lamb, D. W. (2014). Effect of stripe rust on the yield response of wheat to nitrogen. Crop J. 2, 201–206. doi: 10.1016/j.cj.2014.05.002
Dodds, P. N., and Rathjen, J. P. (2010). Plant immunity: towards an integrated view of plant–pathogen interactions. Nat. Rev. Genet. 11, 539–548. doi: 10.1038/nrg2812
Dong, S., Scagel, C. F., Gheng, L., Fuchigami, L. H., and Rygiewicz, P. (2001). Soil temperature and plant growth stage influence nitrogen uptake and amino acid concentration of apple during early spring growth. Tree Physiol. 21, 541–547. doi: 10.1093/treephys/21.8.541
Dordas, C. (2008). Role of nutrients in controlling plant diseases in sustainable agriculture: a review. Agron. Sustain. Dev. 28, 33–46. doi: 10.1051/agro:2007051
Evans, I., Solberg, E., and Huber, D. M. (2007). “Copper and plant disease,” in Mineral nutrition and plant disease, eds L. E. Datnoff, W. H. Elmer, and D. M. Huber (St Paul, MI: APS Press), 177–188.
Fawe, A., Menzies, J. G., Chérif, M., and Bélanger, R. R. (2001). “Silicon and disease resistance in dicotyledons,” in Silicon in Agriculture, eds L. E. Datnoff, G. H. Snyder, and G. H. Korndörfer (Amsterdam: Elsevier), 159–169. doi: 10.1016/S0928-3420(01)80013-6
Fones, H. N., and Preston, G. M. (2012). Reactive oxygen and oxidative stress tolerance in plant pathogenic Pseudomonas. FEMS Microbiol. Lett. 327, 1–8. doi: 10.1111/j.1574-6968.2011.02449.x
Foyer, C. H., Noctor, G., and Hodges, M. (2011). Respiration and nitrogen assimilation: Targeting associated metabolism as a means to enhance nitrogen use efficiency. J. Exp. Bot. 62, 1467–1482.
Gao, R., Ng, F. K. L., Liu, P., and Wong, S. M. (2012). Hibiscus chloroticringspot virus coat protein upregulates sulfur metabolism genes for enhanced pathogen defense. Mol. Plant Microbe Interact. 25, 1574–1583. doi: 10.1094/MPMI-08-12-0203-R
Graham, D. R. (1983). Effects of nutrients stress on susceptibility of plants to disease with particular reference to the trace elements. Adv. Bot. Res. 10, 221–276. doi: 10.1016/S0065-2296(08)60261-X
Graham, D. R., and Webb, M. J. (1991). “Micronutrients and disease resistance and tolerance in plants,” in Micronutrients in Agriculture, 2nd Edn, eds J. J. Mortvedt, F. R. Cox, L. M. Shuman, and R. M. Welch (Madison, WI: Soil Science Society of America, Inc), 329–370. doi: 10.2136/sssabookser4.2ed.c10
Graham, J. H., Dewdney, M. M., and Younce, H. D. (2011). Comparison of copper formulations for control of citrus canker on “Hamlin” orange. Proc Florida State Hortic Soc. 124, 79–84.
Gupta, N., Debnath, S., Sharma, S., Sharma, P., and Purohit, J. (2017). in Agriculturally Important Microbes for Sustainable Agriculture, eds V. S. Meena, et al. (Singapore: Springer Nature Singapore Pte Ltd), 217–251. doi: 10.1007/978-981-10-5343-6_8
Haas, D., and Défago, G. (2005). Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat. Rev. Microbiol. 3, 307–319. doi: 10.1038/nrmicro1129
Heine, G., Max, J. F. J., Fuhrs, H., Moran-Puente, D. W., Heintz, D., and Horst, W. (2011). Effect of manganese on the resistance of tomato to Pseudocercospora fuligena. J. Plant Nutr. Soil Sci. 174, 827–836.
Helfenstein, J., Pawlowski, M. L., Hill, C. B., Stewart, J., Lagos-Kutz, D., Bowen, C. R., et al. (2015). Zinc deficiency alters soybean susceptibility to pathogens and pests. J. Plant Nutr. Soil Sci. 178, 896–903. doi: 10.1002/jpln.201500146
Hendrix, J. W., Guttman, S. M., and Wightman, D. L. (1969). Cation and sterol effects on growth of Phytophthora parasitica var. nicotianae. Phytopathology 59, 1620–1624.
Hirschi, K. D. (2004). The calcium conundrum both versatile nutrient and specific signal. Plant Physiol. 136, 2438–2442. doi: 10.1104/pp.104.046490
Hoffland, E., Van Beusichem, M. L., and Jeger, M. J. (1999). Nitrogen availability and susceptibility of tomato leaves to Botrytis cinerea. Plant Soil 210, 263–272. doi: 10.1023/A:1004661913224
Holzmueller, E. J., Jose, S., and Jenkins, M. A. (2007). Influence of calcium, potassium, and magnesium on Cornus florida L. density and resistance to dogwood anthracnose. Plant Soil 290, 189–199. doi: 10.1007/s11104-006-9151-y
Huber, D., Römheld, V., and Weinmann, M. (2012). “Relationship between Nutrition, plant diseases and pests,” in Marschner’s mineral nutrition of higher plants, 3rd Edn, ed. P. Marschner (Cambridge, MA: Academic Press), 283–298. doi: 10.1016/B978-0-12-384905-2.00010-8
Huang, H., Nguyen Thi Thu, T., He, X., Gravot, A., Bernillon, S., Ballini, E., et al. (2017). Increase of fungal pathogenicity and role of plant glutamine in nitrogen-induced susceptibility (NIS) to rice blast. Front. Plant Sci. 8:265. doi: 10.3389/fpls.2017.00265
Huber, D. M. (1980). “The role of mineral nutrition in defense,” in Plant Disease, An Advanced Treatise, Volume 5, How Plants Defend Themselves, eds J. G. Horsfall and E. B. Cowling (New York, NY: Academic Press), 381–406. doi: 10.1016/B978-0-12-356405-4.50028-9
Huber, D. M., and Graham, R. D. (1999). “The role of nutrition in crop resistance and tolerance to disease,” in Mineral Nutrition of Crops Fundamental Mechanisms and Implications, ed. Z. Rengel (New York, NY: Food Product Press), 205–226.
Huber, D. M., and Haneklaus, S. (2007). Managing nutrition to control plant disease. Landbauforsch. Volk. 4, 313–322.
Huber, D. M., and Jones, J. B. (2013). The role of magnesium in plant disease. Plant Soil 368, 73–85. doi: 10.1007/s11104-012-1476-0
Hussain, I., Alam, S., Khan, I., Shah, B., and Naeem, A. (2016). Study on the biological control of fusarium wilt of tomato. J. Entomol. Zool. Stud. 4, 525–528.
Jewell, J. B., Sowders, J. M., He, R., Willis, M. A., Gang, D. R., and Tanaka, K. (2019). Extracellular ATP shapes a defence-related transcriptome both independently and along with other defence signaling pathways. Plant Physiol. 179, 1144–1158. doi: 10.1104/pp.18.01301
Keinath, P. A., and Loria, R. (1996). “Management of common scab of potato with plant nutrients,” in Management of diseases with macro- and microelements, ed. W. A. Engelhard (St Paul, MI: APS Press), 152–166.
Khaing, E. E., Ahmad, Z. A. M., Yun, W. M., and Ismail, M. R. (2014). Effects of silicon, copper and zinc applications on sheath blight disease severity on rice. World J Agric Res. 2, 309–314. doi: 10.12691/wjar-2-6-11
Khoshgoftarmanesh, A. H., Kabiri, S., Shariatmadari, H., Sharifnabi, B., and Schulin, R. (2010). Zinc nutrition effect on the tolerance of wheat genotypes to Fusarium root-rot disease in a solution culture experiment. Soil Sci. Plant Nutr. 56, 234–243. doi: 10.1111/j.1747-0765.2009.00441.x
Kieu, N. P., Aznar, A., Segond, D., Rigault, M., Simond-Côte, E., Kunz, C., et al. (2012). Iron deficiency affects plant defense responses and confers resistance to Dickeya dadantii and Botrytis cinerea. Mol. Plant Pathol. 13, 816–827. doi: 10.1111/j.1364-3703.2012.00790.x
Kirkegaard, J. A., Munns, R., James, R. A., and Neate, S. M. (1999). Does water and phosphorus uptake limit leaf growth of rhizoctonia-infected wheat seedlings? Plant Soil 209, 157–166. doi: 10.1023/A:1004374516088
Krupinsky, J. M., Halvorson, A. D., Tanaka, D. L., and Merrill, S. D. (2007). Nitrogen and tillage effects on wheat leaf spot diseases in the northern great plains. Agron. J. 99, 562–569. doi: 10.1094/PDIS.2001.85.7.785
Kruse, C., Jost, R., Lipschis, M., Kopp, B., Hartmann, M., and Hell, R. (2007). Sulfur-enhanced defence: effects of sulfur metabolism, nitrogen supply, and pathogen lifestyle. Plant Biol. 9, 608–619. doi: 10.1055/s-2007-965432
Kunstler, A., Gullner, G., Ádám, A. L., Nagy, J. K., and Kiraly, L. (2020). The versatile roles of sulfur-containing biomolecules in plant defense - A road to disease resistance. Plants 9:1705. doi: 10.3390/plants9121705
Lecompte, F., Ali Abro, M., and Nicot, P. C. (2010). Contrasted responses of Botrytis cinerea isolates developing on tomato plants grown under different nitrogen nutrition regimes. Plant Pathol. 59, 891–899. doi: 10.1111/j.1365-3059.2010.02320.x
Lecourieux, D., Mazars, C., Pauly, N., Ranjeva, R., and Pugin, A. (2002). Analysis and effects of cytosolic free calcium increases in response to elicitors in Nicotiana plumbaginifolia cells. Plant Cell 14, 2627–2641. doi: 10.1105/tpc.005579
Lemanceau, P., and Alabouvette, C. (2008). Suppression of fusarium wilts by fluorescent pseudomonads: Mechanisms and applications. Biocontrol Sci. Technol. 3, 219–234. doi: 10.1080/09583159309355278
Lewandowski, I., Hardtlein, M., and Kaltschmitt, M. (1999). Sustainable crop production: definition and methodological approach for assessing and implementing sustainability. Crop Sci. 39, 184–193. doi: 10.2135/cropsci1999.0011183X003900010029x
Li, Z., Fan, Y. C., Gao, L., Cao, X., Ye, J. L., and Li, G. H. (2016). The dual roles of zinc sulfate in mitigating peach gummosis. Plant Dis. 100, 345–351. doi: 10.1094/PDIS-01-15-0131-RE
Long, D. H., Lee, F. N., and TeBeest, D. O. (2000). Effect of nitrogen fertilization on disease progress of rice blast on susceptible and resistant cultivars. Plant Dis. 84, 403–409. doi: 10.1094/PDIS.2000.84.4.403
Lukac, M., Calfapietra, C., Lagomarsino, A., and Loreto, F. (2011). Global climate change and tree nutrition: effects of elevated CO2 and temperature. Tree Physiol. 30, 1209–1220. doi: 10.1093/treephys/tpq040
Machado, P. P., Steiner, F., Zuffo, A. M., and Machado, R. A. (2018). Could the supply of boron and zinc improve resistance of potato to early blight? Potato Res. 61, 169–182. doi: 10.1007/s11540-018-9365-4
Marschner, H., and Marschner, P. (2012). Marschner’s Mineral Nutrition of Higher Plants, 3rd Edn. San Diego, CA: Elsevier Academic Press, 1–651.
Marschner, P. (2012). Marschner’s Mineral Nutrition of Higher Plants, 3rd Edn. London: Academic Press, 178–189.
Meena, V. S., Meena, S. K., Verma, J. P., Meena, R. S., and Ghosh, B. N. (2015). The needs of nutrient use efficiency for sustainable agriculture. J Clean Prod. 102, 562–563. doi: 10.1016/j.jclepro.2015.04.044
Mengel, K. (2001). Principles of Plant Nutrition, 5th Edn. Dordrecht: Kluwer Academic Publishers, 481–509. doi: 10.1007/978-94-010-1009-2_10
Moreira, W. R., Bispo, W. M. S., Rios, J. A., Debona, D., Nascimento, C. W. A., and Rodrigues, F. A. (2015). Magnesium induced alterations in the photosynthetic performance and resistance of the plants infected with Bipolaris oryzae. Sci. Agric. 72, 328–333. doi: 10.1590/0103-9016-2014-0312
Mur, L. A., Simpson, C., Kumari, A., Gupta, A. K., and Gupta, K. J. (2017). Moving nitrogen to the centre of plant defence against pathogens. Ann. Bot. 119, 703–709. doi: 10.1093/aob/mcw179
Najihah, N. I., Hanafi, M. M., Idris, A. S., and Hakim, M. A. (2015). Silicon treatment in oil palms confers resistance to basal stem rot disease caused by Ganoderma boninense. Crop Prot. 67, 151–159. doi: 10.1016/j.cropro.2014.10.004
Nam, M. H., Jeong, S. K., Lee, Y. S., Choi, J. M., and Kim, H. G. (2006). Effects of nitrogen, phosphorus, potassium and calcium nutrition on strawberry anthracnose. Plant Pathol. 55, 246–249. doi: 10.1111/j.1365-3059.2006.01322.x
Neumann, S., Paveley, N. D., Beed, F. D., and Sylvester-Bradley, R. (2004). Nitrogen per unit leaf area affects the upper asymptote of Puccinia striiformis f. sp. tritici epidemics in winter wheat. Plant Pathol. 53, 725–732. doi: 10.1111/j.1365-3059.2004.01107.x
Ngadze, E., Icishahayo, D., Coutinho, T. A., and van der Waals, J. E. (2012). Role of polyphenol oxidase, peroxidase, phenylalanine ammonia lyase, chlorogenic acid, and total soluble phenols in resistance of potatoes to soft rot. Plant Dis. 96, 186–192. doi: 10.1094/PDIS-02-11-0149
Nome, C., Magalhaes, P. C., Oliveira, E., Nome, S., and Laguna, I. G. (2009). Differences in intracellular localization of corn stunt spiroplasmas in magnesium treated maize. Biocell 33, 133–136. doi: 10.32604/biocell.2009.33.133
Nwachukwu, I. D., Slusarenko, A. J., and Gruhlke, M. C. H. (2012). Sulfur and sulfur compounds in plant defence. Nat. Prod. Commun. 7, 395–400. doi: 10.1177/1934578X1200700323
Ojha, R. K., and Jha, S. K. (2021). “Role of mineral nutrition in management of plant diseases,” in Farmers’ Prosperity through Improved Agricultural Technologies, eds H. K. Singh, S. S. Solankey, and M. K. Roy (New Delhi: Jaya Publishing House), 241–261.
Oliveira, E., Oliveira, C., Souza, I., Magalhaes, P., Andrade, C., and Hogenhout, S. (2005). Spiroplasma and phytoplasma reduce kernel production and nutrient and water contents of several but not all maize cultivars. Maydica 50, 171–178.
Oliveira, J. C., Albuquerque, G. M. R., Mariano, R. L. R., Gondim, D. M. F., Oliveira, J. T. A., and Souza, E. B. (2012). Reduction of the severity of angular leaf spot of cotton mediated by silicon. J. Plant Pathol. 94, 297–300.
Oosterhuis, D., Loka, D., Kawakami, E., and Pettigrew, W. (2014). The physiology of potassium in crop production. Adv. Agron. 126, 203–234. doi: 10.1016/B978-0-12-800132-5.00003-1
Osuna-Canizalez, F. J., Datta, S. K., and Bonman, J. M. (1991). Nitrogen form and silicon nutrition effects on resistance to blast disease of rice. Plant Soil 135, 223–231. doi: 10.1007/BF00010910
Pérez-García, A., Snoeijers, S. S., Joosten, M. H., Goosen, T., and De Wit, P. J. (2001). Expression of the avirulence gene Avr9 of the fungal tomato pathogen Cladosporium fulvum is regulated by the global nitrogen response factor NRF1. Mol. Plant Microbe Interact. 14, 316–325. doi: 10.1094/MPMI.2001.14.3.316
Pettigrew, W. T. (2008). Potassium influences on yield and quality production for maize, wheat, soybean and cotton. Physiol. Plantarum. 133, 670–681. doi: 10.1111/j.1399-3054.2008.01073.x
Potash and Phosphate Institute [PPI] (1988). “Phosphorus nutrition improves plant disease resistance,” in Better Crops With Plant Food, ed. PPI (Atlanta, GA: PPI), 22–23.
Pozza, E. A., Pozza, A. A. A., and Botelho, D. M. S. (2015). Silicon in plant disease control. Rev Ceres 62, 323–331. doi: 10.1590/0034-737X201562030013
Prabhu, A. S., Fageria, N. K., and Huber, D. M. (2007). “Potassium nutrition and plant diseases,” in Mineral Nutrition and Plant Disease, eds L. E. Datnoff, W. H. Elmer, and D. M. Huber (Saint Paul, MN: American Phytopathological Society), 57–78.
Prasad, D., Singh, R., and Singh, A. (2010). Management of sheath blight of rice with integrated nutrients. Indian Phytopathol. 63, 11–15.
Reuveni, M., Oppernheim, D., and Reuveni, R. (1998). Integrated control of powdery mildew on apple trees by foliar sprays of mono-potassium phosphate fertilizer and sterol inhibiting fungicides. Crop Prot. 17, 563–568. doi: 10.1016/S0261-2194(98)00050-7
Reuveni, R., Dor, G., Raviv, M., Reuveni, M., and Tuzun, S. (2000). Systemic resistance against Sphaerotheca fuliginea in cucumber plants exposed to phosphate in hydroponics system, and its control by foliar spray of mono-potassium phosphate. Crop Prot. 19, 355–361. doi: 10.1016/S0261-2194(00)00029-6
Roblin, G., Octave, S., Faucher, M., Fleurat-Lessard, P., and Berjeaud, J. M. (2018). Cysteine: A multifaceted amino acid involved in signaling, plant resistance and antifungal development. Plant Physiol. Biochem. 129, 77–89. doi: 10.1016/j.plaphy.2018.05.024
Rolshausen, P. E., and Gubler, W. D. (2005). Use of boron for the control of Eutypa dieback of grapevines. Plant Dis. 89, 734–738. doi: 10.1094/PD-89-0734
Sanjeev, K. K., and Eswaran, A. (2008). Efficacy of micro nutrients on banana fusarium wilt (Fusarium oxysporum f. sp. cubense) and it’s synergistic action with Trichoderma viride. Notulae Botanicae Horti Agro botanici Cluj-Napoca 36, 52–54.
Sarosh, B. R., Sivaramakrishnan, S., and Shetty, H. S. (2005). Elicitation of defense related enzymes and resistance by l-methionine in pearl millet against downy mildew disease caused by Sclerospora graminicola. Plant Physiol. Biochem. 43, 808–815.
Scheible, W.-R., Morcuende, R., Czechowski, T., Fritz, C., Osuna, D., Palacios-Rojas, N., et al. (2004). Genome-wide reprogramming of primary and secondary metabolism, protein synthesis, cellular growth processes, and the regulatory infrastructure of Arabidopsis in response to nitrogen. Plant Physiol. 136, 2483–2499. doi: 10.1104/pp.104.047019
Schurt, D. A., Lopes, U. P., Duarte, H. S. S., and Rodrigues, F. A. (2015). Rice resistance to sheath blight mediated by potassium. J. Phytopathol. 163, 310–313. doi: 10.1111/jph.12269
Shabala, S., and Pottosin, I. I. (2010). Potassium and potassium-permeable channels in plant salt tolerance. Signal. Commun. Plants 2010: 87–110. doi: 10.1007/978-3-642-10494-7_5
Sharma, S., Duveiller, E., Basnet, R., Karki, C. B., and Sharma, R. C. (2005). Effect of potash fertilization on Helminthosporium leaf blight severity in wheat, and associated increases in grain yield and kernel weight. Field Crop Res. 93, 142–150. doi: 10.1016/j.fcr.2004.09.016
Shetty, R., Frette, X., Jensen, B., Shetty, N. P., Jensen, J. D., Jorgensen, H. J. L., et al. (2011). Silicon-induced changes in antifungal phenolic acids, flavonoids, and key phenylpropanoid pathway genes during the interaction between miniature roses and the biotrophic pathogen Podosphaera pannosa. Plant Physiol. 157, 2194–2205. doi: 10.1104/pp.111.185215
Shoemaker, P. B. (1992). Evaluation of different materials for tomato bacterial canker. Fungic Nat.i TEST 47:155.
Snyder, C. S., and Ashlock, L. O. (1996). Late-season potassium deficiency symptoms in southern soybeans. Better Crop 80, 10–11.
Sommer, A. L. (1931). Copper as an essential for plant growth. Plant Physiol. 6, 339–345. doi: 10.1104/pp.6.2.339
Streeter, T. C., Rengel, Z., Neate, S. M., and Graham, R. D. (2001). Zinc fertilization increases tolerance to Rhizoctonia solani (AG 8) in Medicago truncatula. Plant Soil 228, 233–242. doi: 10.1023/A:1004874027331
Sugimoto, T., Watanabe, K., Yoshida, S., Aino, M., Irie, K., Matoh, T., et al. (2011). Select calcium compounds reduce the severity of Phytophthora stem rot of soybean. Plant Dis. 92, 1559–1565. doi: 10.1094/PDIS-92-11-1559
Sun, Q., Liu, X., Yang, J., Liu, W., Du, Q., Wang, H., et al. (2018). MicroRNA528 affects lodging resistance of maize by regulating lignin biosynthesis under nitrogen-luxury conditions. Mol. Plant 11, 806–814. doi: 10.1016/j.molp.2018.03.013
Sun, W., Zhang, J., Fan, Q., Xue, G., Li, Z., and Liang, Y. (2010). Silicon-enhanced resistance to rice blast is attributed to silicon-mediated defense resistance and its role as physical barrier. Eur. J. Plant Pathol. 128, 39–49. doi: 10.1007/s10658-010-9625-x
Sun, Y., Wang, M., Mur, Luis, A., Shen, Q., and Guo, S. (2020). Unravelling the roles of nitrogen nutrition in plant disease defences international journal of molecular sciences. Int. J. Mol. Sci. 21:572. doi: 10.3390/ijms21020572
Sweeney, D. W., Granade, G. V., Eversmeyer, M. G., and Whitney, D. A. (2000). Phosphorus, potassium, chloride, and fungicide effects on wheat yield and leaf rust severity. J. Plant. Nutr. 23, 1267–1281. doi: 10.1080/01904160009382099
Tanaka, K., Choi, J., Cao, Y., and Stacey, G. (2014). Extracellular ATP acts as a damage-associated molecular pattern (DAMP) signal in plants. Front. Plant Sci. 5:446. doi: 10.3389/fpls.2014.00446
Thomashow, L. S. (1996). Biological control of plant root pathogens. Curr. Opin. Biotech. 7, 343–347. doi: 10.1016/S0958-1669(96)80042-5
Thomma, B. P., Van Esse, H. P., Crous, P. W., and De Wit, P. J. (2005). Cladosporium fulvum (syn. Passalora fulva), a highly specialized plant pathogen as a model for functional studies on plant pathogenic Mycosphaerellaceae. Mol. Plant Pathol. 6, 379–393. doi: 10.1111/j.1364-3703.2005.00292.x
Tripathi, D., Zhang, T., Koo, A. J., Stacey, G., and Tanaka, K. (2018). Extracellular ATP acts on jasmonate signaling to reinforce plant defence. Plant Physiol. 176, 511–523. doi: 10.1104/pp.17.01477
Velasquez, A. C., Castroverde, C., and He, S. Y. (2018). Plant-pathogen warfare under changing climate conditions. Curr. biol. 28, 619–634. doi: 10.1016/j.cub.2018.03.054
Walters, D. R., and Bingham, I. J. (2007). Influence of nutrition on disease development caused by fungal pathogens: implications for plant disease control. Ann. App. Biol. 151, 307–324. doi: 10.1111/j.1744-7348.2007.00176.x
Wang, Z., Hassan, M. U., Nadeem, F., Wu, L., Zhang, F., and Li, X. (2020). Magnesium fertilization improves crop yield in most production systems: A meta-analysis. Front. Plant Sci. 10:1727. doi: 10.3389/fpls.2019.01727
Wang, M., Zheng, Q., Shen, Q., and Guo, S. (2013). The critical role of potassium in plant stress response. Int. J. Mol. Sci 14, 7370–7390. doi: 10.3390/ijms14047370
Wang, Y., Loake, G. J., and Chu, C. (2013). Cross-talk of nitric oxide and reactive oxygen species in plant programed cell death. Front. Plant Sci. 4:314. doi: 10.3389/fpls.2013.00314
Wen, Z. H., Li, H. G., Shen, J. B., and Rengel, Z. (2017). Maize responds to low shoot P concentration by altering root morphology rather than increasing root exudation. Plant Soil 416, 377–389. doi: 10.1007/s11104-017-3214-0
White, P. J., and Broadley, M. R. (2003). Calcium in plants. Ann. Bot. 92, 487–511. doi: 10.1093/aob/mcg164
Woltz, S. S., and Jones, J. P. (1979). Effects of magnesium on bacterial spot of pepper and tomato and on the in vitro inhibition of Xanthomonas vesicatoria by streptomycin. Plant Dis. Rep. 63, 182–184.
Young, C. C., Cheng, K. T., and Waller, G. R. (1991). Phenolic compounds in conducive and suppressive soils on clubroot disease of crucifers. Soil Biol. Biochem. 23, 1183–1189.
Keywords: mineral nutrition, disease management, plant growth, nutrient signaling, nutrient use efficiency
Citation: Tripathi R, Tewari R, Singh KP, Keswani C, Minkina T, Srivastava AK, De Corato U and Sansinenea E (2022) Plant mineral nutrition and disease resistance: A significant linkage for sustainable crop protection. Front. Plant Sci. 13:883970. doi: 10.3389/fpls.2022.883970
Received: 25 February 2022; Accepted: 05 August 2022;
Published: 20 October 2022.
Edited by:
Kapuganti Jagadis Gupta, National Institute of Plant Genome Research (NIPGR), IndiaReviewed by:
Olubukola Oluranti Babalola, North-West University, South AfricaCopyright © 2022 Tripathi, Tewari, Singh, Keswani, Minkina, Srivastava, De Corato and Sansinenea. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ruchi Tripathi, assidious.rt35685@gmail.com; apprart@gbpuat.ac.in
 Ruchi Tripathi
Ruchi Tripathi Rashmi Tewari
Rashmi Tewari K. P. Singh1
K. P. Singh1 Chetan Keswani
Chetan Keswani Tatiana Minkina
Tatiana Minkina Anoop Kumar Srivastava
Anoop Kumar Srivastava Ugo De Corato
Ugo De Corato Estibaliz Sansinenea
Estibaliz Sansinenea